Written by Ricardo Felisberto DVM MRCVS Anesthesia Resident
Rex, a 5-year-old male, 17.4kg Springer Spaniel was referred to the Internal Medicine service at SCVS for investigation of pyothorax. On clinical examination, Rex was lethargic, with mild tachypnoea and bradycardia (HR 52 bpm), and heart and lung sounds were muffled on auscultation of the right hemithorax. All other vital parameters were within normal limits.
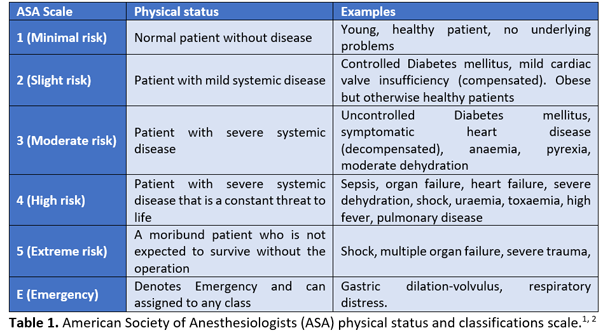
Blood sampling was performed for haematology and biochemistry and was unremarkable. Based on the history and clinical signs, Rex was classified as ASA III (American Society of Anesthesiologists physical status classification system), which represents a “patient with severe systemic disease that limits normal function (compensated)” (Table 1).
Advanced imaging (computed tomography (CT)) was scheduled to investigate the cause of the pyothorax. Rex was premedicated intravenously (IV) with methadone, 0.3mg kg-1, (Comfortan® 10mg/ml, Dechra, UK) and acepromazine, 0.01mg kg-1, (ACP injection® 2mg/ml, Novartis, UK), which produced moderate sedation. Anaesthesia was induced with IV alfaxalone, 1.5 mg kg-1, (Alfaxan 10mg/ml, Jurox, UK), and the trachea intubated with a cuffed 10.5 mm Murphy endotracheal tube. The CT scan revealed a 4cm mass in the mediastinum associated with mild pleural effusion, and it was decided to proceed with a median sternotomy.
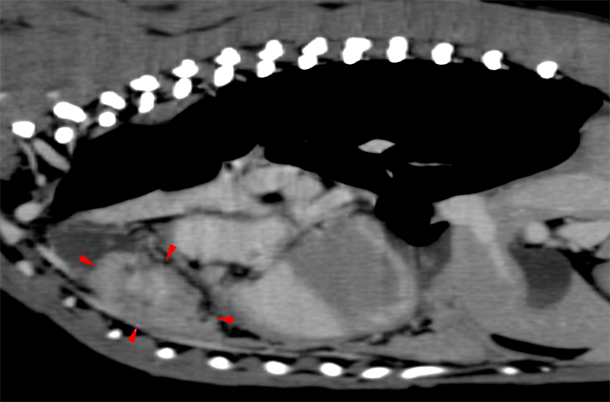
The CT scan revealed a 4cm mass
Rex was already receiving non-steroidal treatment with oral meloxicam, 0.1 mg kg-1, every 24h (Metacam® 1.5mg/ml oral suspension for dogs, Boehringer Ingelheim, UK), and paracetamol, 10mg kg-1, IV every 12h (Perfalgan® 10mg/ml Solution for infusion, Bristol-Myers Squibb Pharmaceuticals limited, UK). To improve intra- and post-operative analgesia, a lumbosacral epidural was performed with preservative-free morphine 1%, 0.1 mg kg-1, (Morphine Sulphate Injection 10 mg/ml, Hameln Pharmaceuticals, ltd, UK) diluted in 0.3ml kg-1 of 0.9% saline (Vetivex® Veterinary 0.9% Sodium Chloride, injection, USP, UK) to facilitate cranial migration of the morphine and provide thoracic region analgesia. A cannula was placed in the dorsal pedal artery for continuous invasive blood pressure monitoring and blood sampling during surgery if required.
Rex was moved into theatre and was connected to a multiparametric monitor (Datex Ohmeda S/5), with electrocardiography, pulse oximetry, capnography, non-invasive and invasive blood pressure, temperature, spirometry and inspired and end-tidal anaesthetic and oxygen concentrations. Variable rate infusions of fentanyl, 0.2 - 0.4 µg kg min-1, (Fentadon 50micrograms/ml, Dechra, UK) and ketamine, 5-10 µg kg min-1, (Anesketin 100mg/ml, Dechra, UK) were started, and IV paracetamol, 10 mg kg-1, infused over 30 minutes (Perfalgan® 10mg/ml Solution for infusion, Bristol-Myers Squibb Pharmaceuticals limited, UK). A peripheral nerve stimulator (Innervator® 252, Fisher&Paykel Healthcare) was placed across the ulnar nerve on the medial aspect of the elbow to monitor neuromuscular function. Prior to surgery commencing, IV atracurium, 0.3mg kg-1, (Atracurium besilate 10mg/ml, Hameln Pharmaceuticals, ltd, UK) was administered to produce neuromuscular blockade, and mechanical ventilation was started and adjusted according to end-tidal carbon dioxide (CO2) levels. The neuromuscular block was monitored every 5 minutes using the nerve stimulator in a train-of-four pattern.
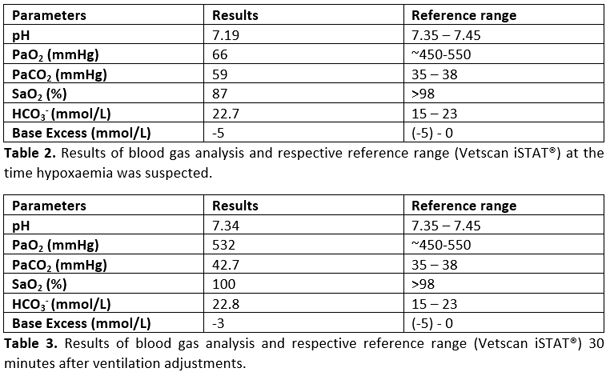
Rex’s anaesthetic was stable until fifty minutes in to the surgery, when the pulse oximeter began to consistently display an SpO2 between 85% to 89%, which persisted even after moving the probe to another place on the tongue. At this point, the end-tidal carbon dioxide concentration was 42 mmHg, and all the remaining parameters were within normal limits. An arterial blood sample was taken for blood gas analysis (Table 2). In line with the pulse oximeter reading, the results confirmed hypoxaemia (low oxygen concentration), and also demonstrated acidaemia (low pH) due to respiratory acidosis (increased CO2) as a consequence of hypoventilation. Consequently, the ventilator settings were adjusted to increase the respiratory rate from 16-22 breaths per minute, and tidal volume from 10ml kg-1 to 15ml kg-1 to increase ventilation/reduce the PaCO2 (which would also increase the pH). Positive end-expiratory pressure (PEEP) was also increased from 4cmH2O to 8cmH2O, which increased the peak inspiratory pressure from 16 to 22cmH2O. Following these adjustments, the SpO2 rapidly increased to 98%, and 30 minutes later, another arterial blood sample was taken (Table 3). The results showed that the increased minute volume from the ventilator had reduced the PaCO2 closer to the reference range, which had also resulted in an increase in the pH towards normal. The hypoxaemia had also resolved, and the oxygen had returned to normal.
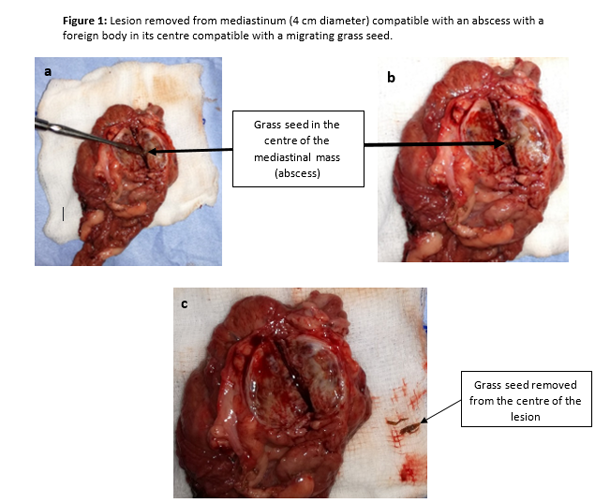
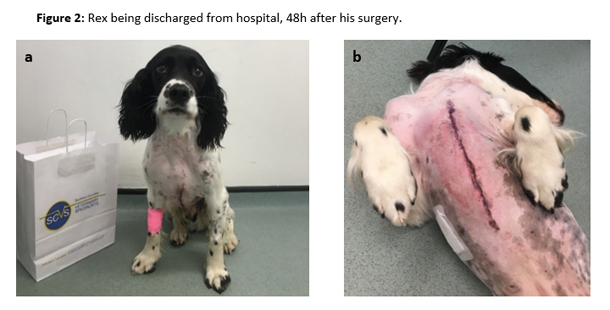
After this event, the patient was stable once again, and the surgeons were able to remove the lesion and perform a subtotal pericardiectomy due to the pericardium involvement. Subsequent dissection of the excised mass revealed an encapsulated grass seed (Figure 1 a, b and c). Rex recovered smoothly and uneventfully from the anaesthetic, and was transferred to ICU where oxygen supplementation was provided via nasal prongs (1.5L min-1) and analgesia was maintained with IV paracetamol, 10mg kg-1 every 12h (Perfalgan® 10mg/ml Solution for infusion, Bristol-Myers Squibb Pharmaceuticals limited, UK), oral meloxicam, 0.1 mg kg-1 every 24h (Metacam® 1.5mg/ml oral suspension for dogs, Boehringer Ingelheim, UK) and an IV methadone (Comfortan® 10mg/ml, Dechra, UK) variable rate infusion between 0.1-0.2 mg kg h-1. Rex was comfortable during the post-operative period, and was discharged from hospital 48h hours after surgery (Figure 2 a, b).
In this particular case, the hypoxaemia was most likely caused by atelectasis (lung collapse), due both to the pre-existing pyothorax, and also packing off of the lungs by the surgeons during thoracic exploration and dissection. The hypercapnia was not indicated by the capnograph, and only detected on arterial blood gas analysis; this discrepancy between the ETCO2 and PaCO2 values was most likely caused by increased alveolar dead space. Alveolar dead space is created by well ventilated but non-perfused alveoli; these alveoli won’t participate in gas exchange with the blood since no blood is reaching the alveolus, and during exhalation, the content of these alveoli will “dilute” the CO2 coming from alveoli where gas exchange has taken place, resulting in a lower ETCO2 reading than the actual PaCO2. The acidaemia was a consequence of the respiratory acidosis (hypercapnia due to hypoventilation). The atelectatic areas of the lung were recruited by increasing the tidal volume and PEEP on the ventilator settings which would have helped to reduce the number of closed alveoli, thus improving oxygen and carbon dioxide exchange in these previously collapsed areas. In conjunction with the increased tidal volume, the increase in respiratory rate helped to further increase the minute respiratory volume of the patient, which contributed to resolution of the hypercapnia and the acidaemia. The use of PEEP during positive pressure ventilation (PPV) helps to maintain pressure in the airway during expiration, which will hold the lung in a semi-inflated state and can also help ‘open up’ previously collapsed alveoli to improve lung-oxygenating ability (Canfrán S, 2012; Hopper K, 2013). The downside of using PEEP is the risk of increasing the peak airway pressure, which can contribute to barotrauma (pressure trauma to the lung), and compromise venous return to the heart, which can reduce cardiac output, leading to hypotension. For these reasons, when utilising PPV with PEEP, it is imperative to intensively monitor both ventilation and haemodynamics (e.g. continuous spirometry, capnography, pulse oximetry, arterial blood pressure measurement and intermittent blood gas analysis if any concerns are raised) (Hopper K, 2013).
Although this was a reasonably straightforward thoracic surgery compared to many that we perform at SCVS, it did throw up some challenges, necessitating clear and continuous communication between the Anaesthesia and Soft Tissue teams. Our ability to provide more complex modes of mechanical ventilation to facilitate adequate oxygenation, as well as having access to invasive monitoring and arterial blood gas analysis, undoubtedly helped us achieve a successful outcome for Rex.